2014-2015 Influenza Season Week 48 ending November 29, 2014
All data are preliminary and may change as more reports are received.
Synopsis:
During week 48 (November 23-29, 2014), influenza activity increased in the United States.
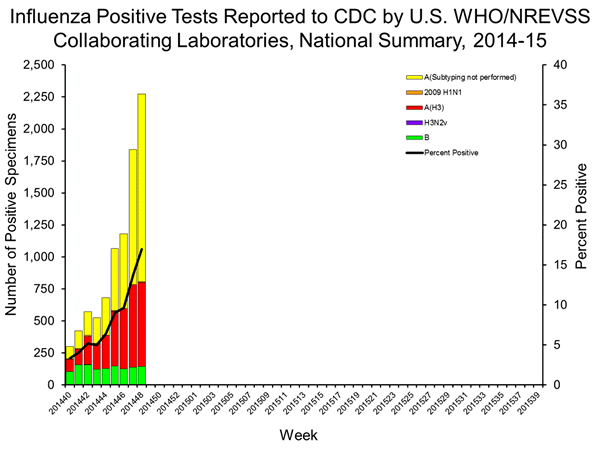
- Viral Surveillance: Of 13,398 specimens tested and reported by U.S. World Health Organization (WHO) and National Respiratory and Enteric Virus Surveillance System (NREVSS) collaborating laboratories during week 48, 2,274 (17.0%) were positive for influenza.
- Pneumonia and Influenza Mortality: The proportion of deaths attributed to pneumonia and influenza (P&I) was below the epidemic threshold.
- Influenza-associated Pediatric Deaths: No influenza-associated pediatric deaths were reported.
- Influenza-associated Hospitalizations: A cumulative rate for the season of 1.8 laboratory-confirmed influenza-associated hospitalizations per 100,000 population was reported.
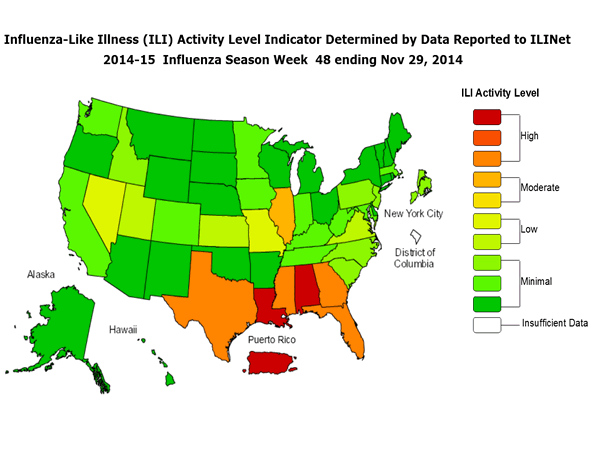
- Outpatient Illness Surveillance:The proportion of outpatient visits for influenza-like illness (ILI) was 2.6%, above the national baseline of 2.0%. Eight of 10 regions reported ILI at or above region-specific baseline levels. Puerto Rico and six states experienced high ILI activity; one state experienced moderate ILI activity; five states experienced low ILI activity; New York City and 38 states experienced minimal ILI activity; and the District of Columbia had insufficient data.
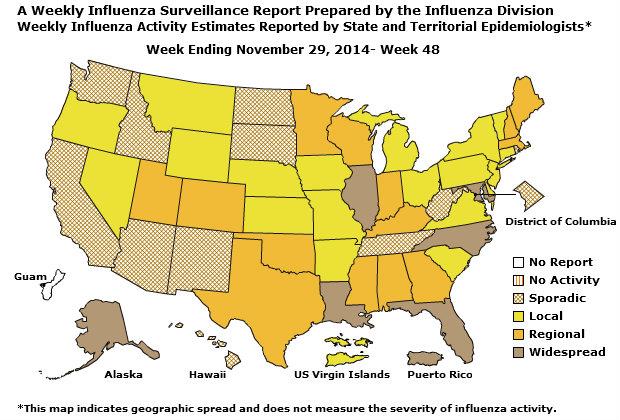
- Geographic Spread of Influenza: The geographic spread of influenza in Puerto Rico and six states was reported as widespread; 14 states reported regional activity; the U.S. Virgin Islands and 19 states reported local activity; the District of Columbia and 11 states reported sporadic activity; and Guam did not report.
| HHS Surveillance Regions* | Data cumulative since September 28, 2014 (week 40) | |||||||
|---|---|---|---|---|---|---|---|---|
| Out-patient ILI† | Number of jurisdictions reporting regional or widespread activity§ | % Respiratory specimens positive for flu‡ | A(H1N1)pdm09 | A (H3) | A(Subtyping not performed) | B | Pediatric Deaths | |
| Nation | Elevated | 21 of 54 | 17.0% | 33 | 3,093 | 4,502 | 1,234 | 5 |
| Region 1 | Elevated | 3 of 6 | 2.3% | 1 | 36 | 24 | 15 | 0 |
| Region 2 | Elevated | 1 of 4 | 1.7% | 6 | 114 | 33 | 29 | 0 |
| Region 3 | Elevated | 1 of 6 | 8.5% | 1 | 223 | 131 | 44 | 0 |
| Region 4 | Elevated | 6 of 8 | 14.1% | 2 | 484 | 2,039 | 646 | 4 |
| Region 5 | Elevated | 4 of 6 | 19.8% | 9 | 473 | 831 | 66 | 0 |
| Region 6 | Elevated | 3 of 5 | 22.7% | 4 | 569 | 1,116 | 296 | 0 |
| Region 7 | Elevated | 0 of 4 | 10.0% | 2 | 203 | 63 | 42 | 0 |
| Region 8 | Normal | 2 of 6 | 11.2% | 3 | 233 | 131 | 27 | 0 |
| Region 9 | Normal | 0 of 5 | 3.0% | 4 | 164 | 66 | 51 | 1 |
| Region 10 | Elevated | 1 of 4 | 18.4% | 1 | 594 | 68 | 18 | 0 |
*HHS regions (Region 1 CT, ME, MA, NH, RI, VT; Region 2: NJ, NY, Puerto Rico, US Virgin Islands; Region 3: DE, DC, MD, PA, VA, WV; Region 4: AL, FL, GA, KY, MS, NC, SC, TN; Region 5: IL, IN, MI, MN, OH, WI; Region 6: AR, LA, NM, OK, TX; Region 7: IA, KS, MO, NE; Region 8: CO, MT, ND, SD, UT, WY; Region 9: AZ, CA, Guam, HI, NV; and Region 10: AK, ID, OR, WA).
† Elevated means the % of visits for ILI is at or above the national or region-specific baseline
§ Includes all 50 states, the District of Columbia, Guam, Puerto Rico, and U.S. Virgin Islands
‡ National data are for current week; regional data are for the most recent three weeks
U.S. Virologic Surveillance:
WHO and NREVSS collaborating laboratories located in all 50 states, Puerto Rico, and the District of Columbia report to CDC the number of respiratory specimens tested for influenza and the number positive by influenza virus type and influenza A virus subtype. The results of tests performed during the current week are summarized in the table below. Region specific data are available at http://gis.cdc.gov/grasp/fluview/fluportaldashboard.html.
| Week 48 | |
|---|---|
| No. of specimens tested | 13,398 |
| No. of positive specimens (%) | 2,274 (17.0%) |
| Positive specimens by type/subtype | |
| Influenza A | 2,129 (93.6%) |
| A(H1N1)pdm09 | 5 (0.2%) |
| H3 | 656 (30.8%) |
| Subytping not performed | 1,468 (69.0%) |
| Influenza B | 145 (6.4%) |
View National and Regional Level Graphs and Data | View Chart Data | View Full Screen | View PowerPoint Presentation
Influenza Virus Characterization*:
CDC has characterized 132 influenza viruses [1 A(H1N1)pdm09, 114 A(H3N2), and 17 influenza B viruses] collected by U.S. laboratories since October 1, 2014.
Influenza A Virus [115]
- A(H1N1)pdm09 [1]: The one H1N1 virus tested was characterized as A/California/7/2009-like, the influenza A (H1N1) component of the 2014-2015 Northern Hemisphere influenza vaccine.
- A (H3N2) [114]: Forty-eight (42%) of the 114 H3N2 viruses tested have been characterized as A/Texas/50/2012-like, the influenza A (H3N2) component of the 2014-2015 Northern Hemisphere influenza vaccine. Sixty-six (58%) of the 114 viruses tested showed either reduced titers with antiserum produced against A/Texas/50/2012 or belonged to a genetic group that typically shows reduced titers to A/Texas/50/2012. Among viruses that showed reduced titers with antiserum raised against A/Texas/50/2012, most were antigenically similar to A/Switzerland/9715293/2013, the H3N2 virus selected for the 2015 Southern Hemisphere influenza vaccine. A/Switzerland/9715293/2013 is related to, but antigenically and genetically distinguishable, from the A/Texas/50/2012 vaccine virus. A/Switzerland-like H3N2 viruses were first detected in the United States in small numbers in March of 2014 and began to increase through the spring and summer.
Influenza B Virus [17]
Ten (58.8%) of the influenza B viruses tested belong to B/Yamagata/16/88 lineage and the remaining seven (41.2%) influenza B viruses tested belong to B/Victoria/02/87 lineage.
- Yamagata Lineage [10]: All ten B/Yamagata-lineage viruses were characterized as B/Massachusetts/2/2012-like, which is included as an influenza B component of the 2014-2015 Northern Hemisphere trivalent and quadrivalent influenza vaccines.
- Victoria Lineage [7]: All seven B/Victoria-lineage viruses were characterized as B/Brisbane/60/2008-like, the virus that is included as an influenza B component of the 2014-2015 Northern Hemisphere quadrivalent influenza vaccine.
*CDC routinely uses hemagglutination inhibition (HI) assays to antigenically characterize influenza viruses year-round to compare how similar currently circulating influenza viruses are to those included in the influenza vaccine, and to monitor for changes in circulating influenza viruses. However, a portion of recent influenza A(H3N2) viruses do not grow to sufficient hemagglutination titers for antigenic characterization by HI. For many of these viruses, CDC is also performing genetic characterization to infer antigenic properties.
Antiviral Resistance:
Testing of influenza A(H1N1)pdm09, A(H3N2), and influenza B virus isolates for resistance to neuraminidase inhibitors (oseltamivir and zanamivir) is performed at CDC using a functional assay. Additional A(H1N1)pdm09 and A(H3N2) clinical samples are tested for mutations of the virus known to confer oseltamivir resistance. The data summarized below combine the results of both testing methods. These samples are routinely obtained for surveillance purposes rather than for diagnostic testing of patients suspected to be infected with antiviral-resistant virus.
High levels of resistance to the adamantanes (amantadine and rimantadine) persist among A(H1N1)pdm09 and A(H3N2) viruses (the adamantanes are not effective against influenza B viruses). Therefore, data from adamantane resistance testing are not presented below.
| Oseltamivir | Zanamivir | |||
|---|---|---|---|---|
| Virus Samples tested (n) | Resistant Viruses, Number (%) | Virus Samples tested (n) | Resistant Viruses, Number (%) | |
| Influenza A (H3N2) | 71 | 0 (0.0) | 71 | 0 (0.0) |
| Influenza B | 24 | 0 (0.0) | 24 | 0 (0.0) |
| A(H1N1)pdm09 | 5 | 0 (0.0) | 5 | 0 (0.0) |
In the United States, all recently circulating influenza viruses have been susceptible to the neuraminidase inhibitor antiviral medications, oseltamivir and zanamivir; however, rare sporadic instances of oseltamivir-resistant A(H1N1)pdm09 and A(H3N2) viruses have been detected worldwide. Antiviral treatment with oseltamivir or zanamivir is recommended as early as possible for patients with confirmed or suspected influenza who have severe, complicated, or progressive illness; who require hospitalization; or who are at high risk for serious influenza-related complications. Additional information on recommendations for treatment and chemoprophylaxis of influenza virus infection with antiviral agents is available at http://www.cdc.gov/flu/antivirals/index.htm.
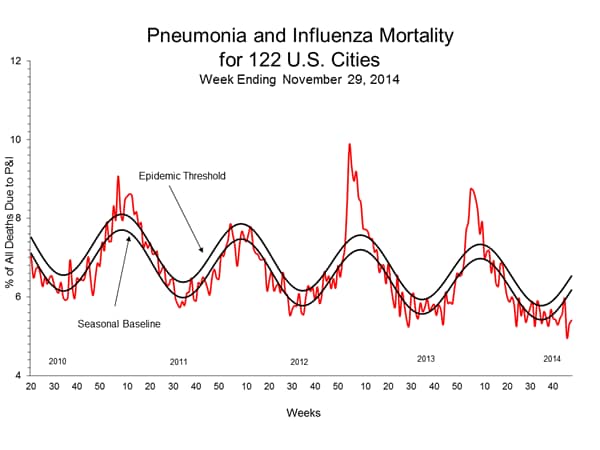
Pneumonia and Influenza (P&I) Mortality Surveillance:
During week 48, 5.4% of all deaths reported through the 122 Cities Mortality Reporting System were due to P&I. This percentage was below the epidemic threshold of 6.5% for week 48.
View Full Screen | View PowerPoint Presentation
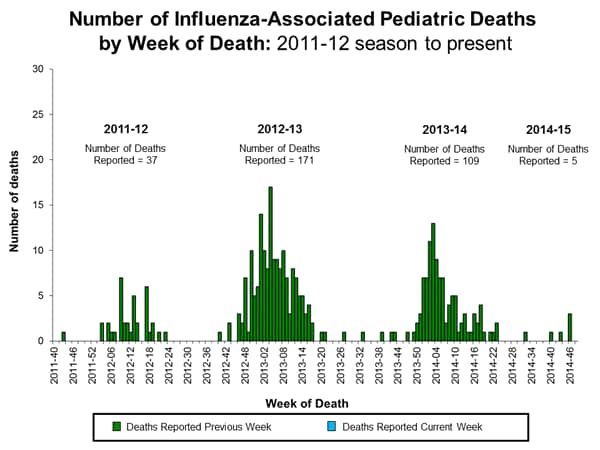
Influenza-Associated Pediatric Mortality:
No influenza-associated pediatric deaths were reported to CDC during week 48. To date, five influenza-associated pediatric deaths have been reported for the 2014-2015 season.
Additional data can be found at: http://gis.cdc.gov/GRASP/Fluview/PedFluDeath.html.
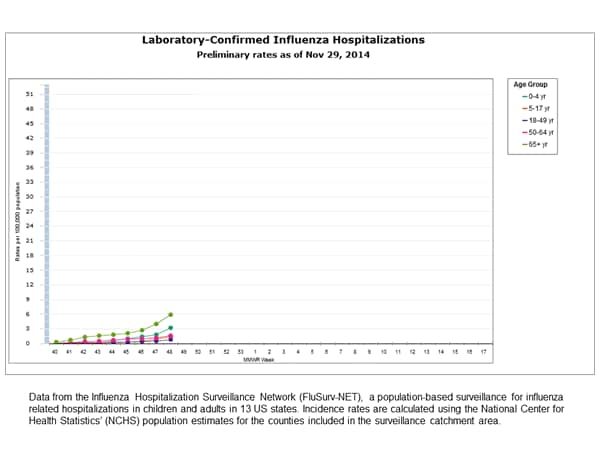
Influenza-Associated Hospitalizations:
The Influenza Hospitalization Surveillance Network (FluSurv-NET) conducts population-based surveillance for laboratory-confirmed influenza-related hospitalizations in children younger than 18 years of age (since the 2003-2004 influenza season) and adults (since the 2005-2006 influenza season).
The FluSurv-NET covers more than 70 counties in the 10 Emerging Infections Program (EIP) states (CA, CO, CT, GA, MD, MN, NM, NY, OR, and TN) and additional Influenza Hospitalization Surveillance Project (IHSP) states. The IHSP began during the 2009-2010 season to enhance surveillance during the 2009 H1N1 pandemic. IHSP sites included IA, ID, MI, OK and SD during the 2009-2010 season; ID, MI, OH, OK, RI, and UT during the 2010-2011 season; MI, OH, RI, and UT during the 2011-2012 season; IA, MI, OH, RI, and UT during the 2012-2013 season; and MI, OH, and UT during the 2013-2014 and 2014-15 seasons.
Data gathered are used to estimate age-specific hospitalization rates on a weekly basis, and describe characteristics of persons hospitalized with severe influenza illness. The rates provided are likely to be an underestimate as influenza-related hospitalizations can be missed, either because testing is not performed, or because cases may be attributed to other causes of pneumonia or other common influenza-related complications.
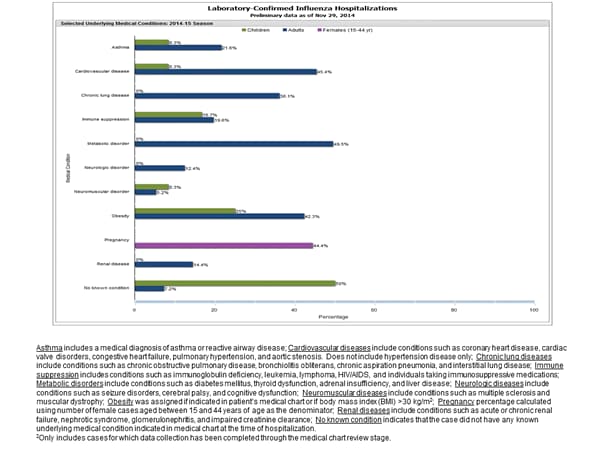
Between October 1, 2014 and November 29, 2014, 500 laboratory-confirmed influenza-associated hospitalizations were reported. The overall hospitalization rate was 1.8 per 100,000 population. The highest rate of hospitalization was among adults aged ≥65 years (5.9 per 100,000 population). Among all hospitalizations, 444 (88.8%) were associated with influenza A, 51 (10.2%) with influenza B, 3 (0.6%) with influenza A and B co-infection, and 2 (0.4%) had no virus type information. Among those with influenza A subtype information, 116 (100%) were H3N2 virus.
Clinical findings are preliminary and based on 109 (22%) pediatric and adult cases with complete medical chart abstraction. The majority (93%) of hospitalized adults had at least one reported underlying medical condition; the most commonly reported were metabolic disorders, cardiovascular disease, and obesity. Seven (7%) hospitalized adults had no identified underlying medical conditions. Among the 12 hospitalized children with complete medical chart abstraction, 6 (50%) had no identified underlying medical conditions. Among the nine hospitalized women of childbearing age (15-44 years), four were pregnant.
Additional FluSurv-NET data can be found at:http://gis.cdc.gov/GRASP/Fluview/FluHospRates.html and http://gis.cdc.gov/grasp/fluview/FluHospChars.html.
View Interactive Application | View Full Screen | View PowerPoint Presentation
Outpatient Illness Surveillance:
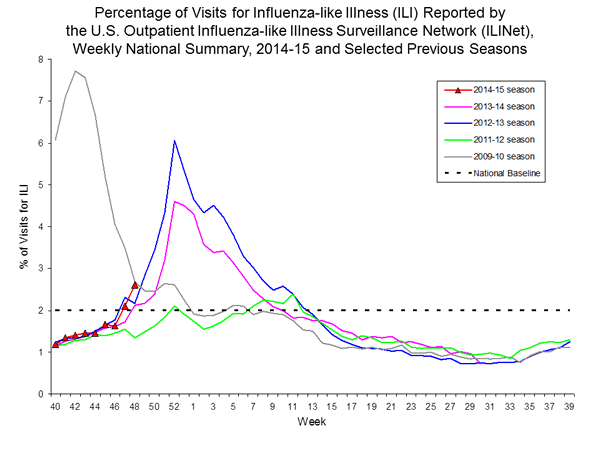
Nationwide during week 48, 2.6% of patient visits reported through the U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet) were due to influenza-like illness (ILI). This percentage is above the national baseline of 2.0%.
(ILI is defined as fever (temperature of 100°F [37.8°C] or greater) and cough and/or sore throat.)
Additional data are available at http://gis.cdc.gov/grasp/fluview/fluportaldashboard.html.
View National and Regional Level Graphs and Data | View Chart Data | View Full Screen | View PowerPoint Presentation
On a regional level, the percentage of outpatient visits for ILI ranged from 1.0% to 4.5% during week 48. Eight regions (Regions 1, 2, 3, 4, 5, 6, 7, and 10) reported a proportion of outpatient visits for ILI at or above their region-specific baseline levels.
ILINet State Activity Indicator Map:
Data collected in ILINet are used to produce a measure of ILI activity* by state. Activity levels are based on the percent of outpatient visits in a state due to ILI and are compared to the average percent of ILI visits that occur during weeks with little or no influenza virus circulation. Activity levels range from minimal, which would correspond to ILI activity from outpatient clinics being below, or only slightly above, the average, to high, which would correspond to ILI activity from outpatient clinics being much higher than average.
During week 48, the following ILI activity levels were experienced:
- Puerto Rico and six states (Alabama, Florida, Georgia, Louisiana, Mississippi, and Texas) experienced high ILI activity.
- One state (Illinois) experienced moderate ILI activity.
- Five states (Kansas, Missouri, Nevada, Utah, and Virginia) experienced low ILI activity.
- New York City and 38 states (Alaska, Arizona, Arkansas, California, Colorado, Connecticut, Delaware, Hawaii, Idaho, Indiana, Iowa, Kentucky, Maine, Maryland, Massachusetts, Michigan, Minnesota, Montana, Nebraska, New Hampshire, New Jersey, New Mexico, New York, North Carolina, North Dakota, Ohio, Oklahoma, Oregon, Pennsylvania, Rhode Island, South Carolina, South Dakota, Tennessee, Vermont, Washington, West Virginia, Wisconsin, and Wyoming) experienced minimal ILI activity.
- Data were insufficient to calculate an ILI activity level from the District of Columbia.
*This map uses the proportion of outpatient visits to health care providers for influenza-like illness to measure the ILI activity level within a state. It does not, however, measure the extent of geographic spread of flu within a state. Therefore, outbreaks occurring in a single city could cause the state to display high activity levels.
Data collected in ILINet may disproportionally represent certain populations within a state, and therefore, may not accurately depict the full picture of influenza activity for the whole state.
Data displayed in this map are based on data collected in ILINet, whereas the State and Territorial flu activity map is based on reports from state and territorial epidemiologists. The data presented in this map is preliminary and may change as more data is received.
Differences in the data presented here by CDC and independently by some state health departments likely represent differing levels of data completeness with data presented by the state likely being the more complete.
Geographic Spread of Influenza as Assessed by State and Territorial Epidemiologists
The influenza activity reported by state and territorial epidemiologists indicates geographic spread of influenza viruses, but does not measure the severity of influenza activity.
During week 48, the following influenza activity was reported:
- Widespread influenza activity was reported by Puerto Rico and six states (Alaska, Florida, Illinois, Louisiana, Maryland, and North Carolina).
- Regional influenza activity was reported by 14 states (Alabama, Colorado, Georgia, Indiana, Kentucky, Maine, Massachusetts, Minnesota, Mississippi, New Hampshire, Oklahoma, Texas, Utah, and Wisconsin).
- Local influenza activity was reported by the U.S. Virgin Islands and 19 states (Arkansas, Connecticut, Delaware, Iowa, Kansas, Michigan, Missouri, Montana, Nebraska, Nevada, New Jersey, New York, Ohio, Oregon, Pennsylvania, South Carolina, Vermont, Virginia, and Wyoming).
- Sporadic influenza activity was reported by the District of Columbia and 11 states (Arizona, California, Hawaii, Idaho, New Mexico, North Dakota, Rhode Island, South Dakota, Tennessee, Washington, and West Virginia).
- Guam did not report.